Interactive PDF Lesson Study Kit
Welcome to your generated study kit! This page is the central hub for all the content created from your document. Use the links in the sidebar to navigate to the different sections.
Home > Formal Lesson Plan
Formal Lesson Plan
Stage 1: Desired Results
- Established Goals:
- Students will understand the fundamental properties and significance of elements, particularly carbon, as outlined in basic chemistry standards.
- Students will comprehend the organization and information provided by the periodic table.
- Students will grasp the concept of chemical bonding, specifically covalent bonding, and its implications for compound diversity.
- Students will recognize the importance of carbon in organic chemistry and the existence of allotropes.
- Understandings: Students will understand that...
- Carbon is a foundational element in the universe with specific atomic characteristics that place it distinctly on the periodic table.
- Carbon's unique capacity to form stable covalent bonds with itself and other elements is the basis for its widespread presence and the vast diversity of organic compounds.
- The same element, carbon, can exist in different structural forms (allotropes), each with distinct properties.
- Carbon's bonding versatility makes it the cornerstone, or "backbone," of organic chemistry.
- Essential Questions:
- What fundamental properties define carbon and distinguish it from other elements?
- How does carbon's unique bonding ability contribute to its prevalence and the complexity of chemical compounds?
- In what ways do carbon's distinct forms (allotropes) exemplify the diversity an element can exhibit?
- Why is carbon considered the "backbone" of organic chemistry, and what are the implications of this role?
- Learning Objectives (Bloom's Taxonomy):
- Remember:
- Students will recall carbon's atomic number (six) and its classification as a non-metal.
- Students will identify carbon's group on the periodic table (Group 14).
- Students will list the three notable allotropes of carbon (graphite, diamond, amorphous carbon).
- Understand:
- Students will explain the concept of carbon's unique ability to form stable covalent bonds with itself and other elements.
- Students will summarize why carbon is considered the "backbone of organic chemistry."
- Students will describe what an allotrope is in the context of carbon's different forms.
- Apply:
- Students will locate carbon on a provided periodic table based on its atomic number and group.
- Students will predict the general type of bonding carbon will undergo based on its classification as a non-metal.
- Analyze:
- Students will differentiate between the implication of carbon forming a "vast array of compounds" versus elements with limited bonding capabilities.
- Students will examine how the concept of allotropy implies structural differences between substances like graphite and diamond.
- Evaluate:
- Students will justify the designation of carbon as a "fundamental element" based on its properties and role in chemistry.
- Students will assess the importance of carbon's unique bonding for the existence of diverse organic molecules.
- Create:
- Students will construct a simple diagram or model representing a carbon atom's potential for covalent bonding with multiple other atoms.
- Students will propose a hypothetical everyday material that might contain carbon, given its properties.
Stage 2: Assessment Evidence
- Performance Tasks:
- Carbon Element Profile: Students will research and create a detailed profile of carbon, including its atomic characteristics, bonding properties, examples of compounds, and its allotropes. This profile must explicitly address its uniqueness and significance as the "backbone of organic chemistry" through a short explanatory paragraph. (Addresses Understand, Apply, Analyze, Evaluate, Create).
- Allotrope Comparison Chart: Students will design a comparison chart for diamond and graphite, outlining their basic structural implications (based on the fact they are allotropes) and correlating them with potential differences in observed properties (e.g., hardness, conductivity, even if specific details are not provided in the source text, the concept of different forms implies different properties). (Addresses Understand, Apply, Analyze).
- Other Evidence:
- Quizzes: Multiple-choice and short-answer questions testing recall of carbon's atomic number, group, classification, and definition of key terms (covalent bond, allotrope).
- Exit Tickets: Students will respond to prompts such as "List three reasons why carbon is a unique element" or "Explain in one sentence why carbon is the backbone of organic chemistry."
- Class Discussions: Active participation and articulation of understanding regarding carbon's bonding and its implications for compound formation.
- Concept Mapping: Students create a concept map connecting "carbon" to its atomic properties, bonding characteristics, resulting compounds, and allotropes.
- Observation: Teacher observation of student engagement, question-asking, and collaborative learning during activities.
Stage 3: Learning Plan
- Learning Activities:
- Engage (10-15 minutes): The Carbon Conundrum
- Activity: Begin with a thought-provoking question: "What do diamonds, pencils, and every living thing on Earth have in common?" Show images of a diamond, a graphite pencil, and a plant.
- Discussion: Facilitate a brief discussion to elicit initial student hypotheses. Introduce carbon as the unifying answer.
- Objective: Hook students' interest and introduce the concept of carbon's diverse forms and importance.
- Explore (15-20 minutes): Carbon's Identity Card
- Activity: Introduce carbon using the provided text. Students will annotate a copy of the text, highlighting key facts: atomic number, non-metal, Group 14, unique bonding.
- Mini-Lecture/Demonstration: Briefly explain the periodic table location and significance of atomic number/group.
- Objective: Students will recall and identify carbon's fundamental atomic characteristics (Bloom's: Remember).
- Explain (25-30 minutes): The Power of Four
- Activity: Focus on the "unique ability to form stable covalent bonds."
- Explain covalent bonding in simple terms (sharing electrons).
- Emphasize carbon's ability to bond with itself and other elements, forming four bonds (tetravalent). Use simple 2D or 3D models (e.g., molecular modeling kit, online simulator) to demonstrate this property.
- Key Concept: Introduce the term "organic chemistry" and explain why carbon's bonding makes it the "backbone."
- Objective: Students will understand and explain carbon's unique bonding properties and its role in organic chemistry (Bloom's: Understand, Apply).
- Elaborate (20-25 minutes): Carbon's Many Faces – Allotropes
- Activity: Introduce the concept of "allotropes" as different structural forms of the same element.
- Examples: Present images and brief descriptions of graphite, diamond, and amorphous carbon. Discuss how their physical properties differ due to their underlying structure (e.g., diamond's hardness vs. graphite's slipperiness, even without detailed atomic structures, students can infer from their known uses).
- Discussion: Lead a discussion on how a single element can manifest in such different ways.
- Objective: Students will list and describe carbon's allotropes and begin to analyze the implications of different structures (Bloom's: Remember, Understand, Analyze).
- Evaluate (10-15 minutes): Synthesizing Carbon's Story
- Activity: Distribute an exit ticket with questions like:
- "What is one unique property of carbon?"
- "Why is carbon considered the 'backbone of organic chemistry'?"
- "Name two allotropes of carbon."
- Homework/Performance Task Introduction: Assign the "Carbon Element Profile" or "Allotrope Comparison Chart" as a longer-term assessment, explaining expectations and resources.
- Objective: Assess immediate recall, understanding, and initial ability to apply concepts (Bloom's: Remember, Understand, Apply, Analyze, Evaluate).
Home > Summary & Key Points
Summary & Key Points
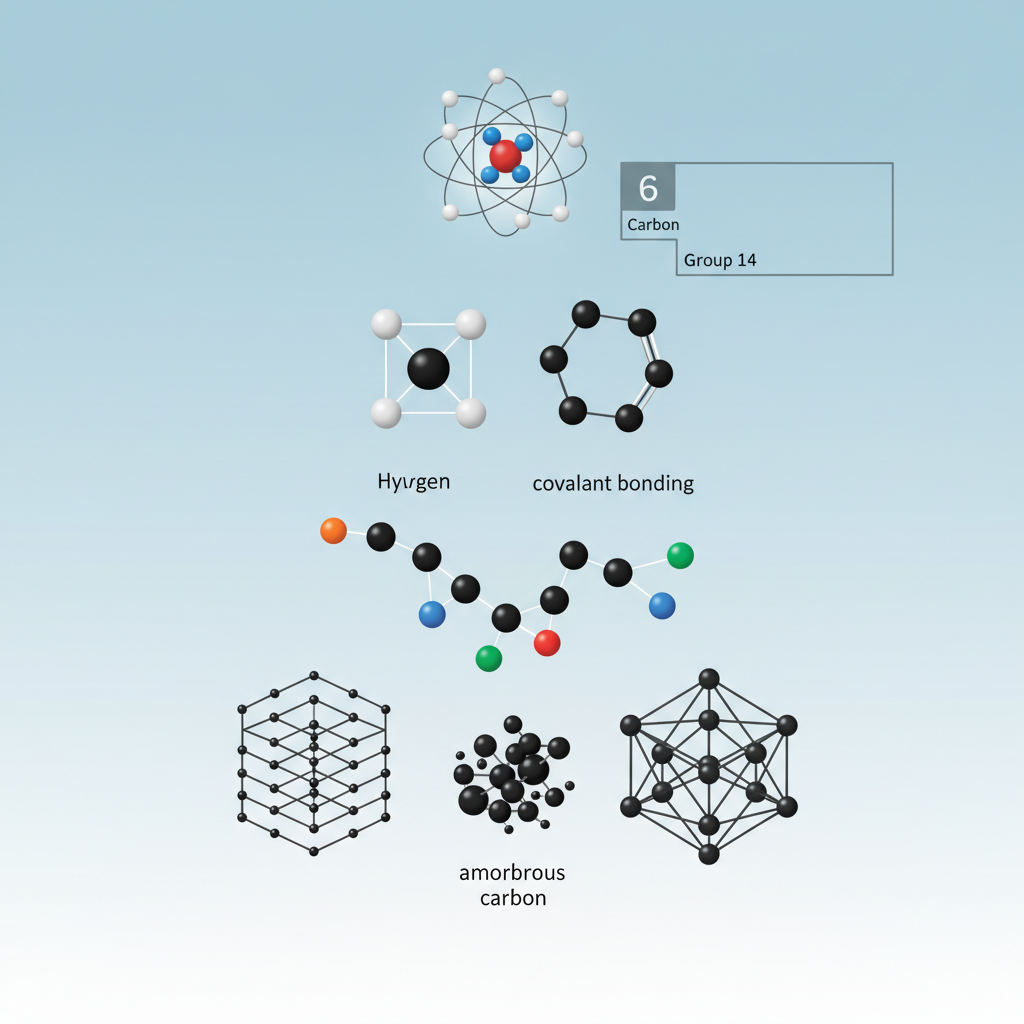
The document describes carbon as a fundamental element with unique properties that make it essential to chemistry. It highlights carbon's atomic characteristics, its distinctive bonding capabilities, and its various forms.
Main Topics:
- Carbon's identity (atomic number, classification, periodic table group)
- Carbon's unique covalent bonding ability
- The role of carbon in forming diverse compounds
- Carbon as the backbone of organic chemistry
- The concept of carbon allotropes (graphite, diamond, amorphous carbon)
Top 5 Key Takeaways:
- Fundamental Identity: Carbon is a fundamental non-metal element, atomic number 6, found in Group 14 of the periodic table.
- Unique Covalent Bonding: It possesses the singular ability to form stable covalent bonds, not only with other elements but significantly, with itself.
- Vast Compound Diversity: This bonding versatility allows carbon to create an immense and diverse array of chemical compounds.
- Organic Chemistry's Backbone: Carbon's unique properties make it the foundational element, or "backbone," of all organic chemistry.
- Allotropic Forms: Carbon exists in multiple distinct structural forms called allotropes, such as graphite, diamond, and amorphous carbon.
Related Videos to Explore
For the point: Unique Covalent Bonding: It possesses the singular ability to form stable covalent bonds, not only with other elements but significantly, with itself.
For the point: Vast Compound Diversity: This bonding versatility allows carbon to create an immense and diverse array of chemical compounds.
See also: Document Outline, Glossary of Terms
Home > Document Outline
Document Outline
- Carbon
- Fundamental Element
- Atomic Number: Six
- Classification: Non-metal
- Periodic Table Group: 14
- Unique Properties
- Ability to Form Stable Covalent Bonds
- With other elements
- With itself
- Results of Bonding Ability
- Forms a vast array of compounds
- Backbone of Organic Chemistry
- Allotropes (Different Forms of Carbon)
- Graphite
- Diamond
- Amorphous Carbon
For a more detailed explanation, see the Detailed Study Guide.
Home > Detailed Study Guide
Detailed Study Guide
This document provides a concise yet rich overview of carbon, a critically important element in chemistry and the natural world. It highlights carbon's fundamental atomic identity, its distinctive bonding behavior, and the profound implications of these properties, including its role as the basis of organic chemistry and its existence in various physical forms.
1. Carbon's Fundamental Identity
Carbon is introduced as a fundamental element in the universe. This signifies its ubiquitous presence and essential role in both inorganic and organic matter. Key atomic identifiers are provided:
- Atomic Number Six: The atomic number defines an element. For carbon, 'six' means every carbon atom has exactly six protons in its nucleus. This number also dictates the number of electrons in a neutral atom, which is crucial for its chemical behavior.
- Non-metal Classification: Carbon belongs to the category of non-metals. Non-metals typically have different physical and chemical properties than metals; they tend to gain or share electrons when forming bonds, rather than losing them.
- Group 14 of the Periodic Table: Its position in Group 14 (also known as the carbon group) provides important clues about its valency – its combining power. Elements in Group 14 typically have four valence electrons, meaning they tend to form four bonds to achieve a stable electron configuration.
2. Unique Covalent Bonding Abilities
The most distinctive and significant property of carbon highlighted in the document is its "ability to form stable covalent bonds with other elements, including itself."
- Covalent Bonds: This refers to a type of chemical bond where atoms share electron pairs. Covalent bonds are generally strong and stable.
- Bonding Versatility:
- With other elements: Carbon readily forms bonds with a wide range of other elements, particularly hydrogen, oxygen, nitrogen, sulfur, and phosphorus. This versatility allows for the construction of incredibly diverse molecular structures.
- With itself: Crucially, carbon atoms can also bond strongly with other carbon atoms. This property, known as catenation, allows carbon to form long chains, branched structures, and rings, which can be single, double, or triple bonds. This self-bonding capability is what sets carbon apart and allows for molecular complexity.
3. Implications of Carbon's Bonding: The Backbone of Organic Chemistry
The unique bonding property has two major consequences, as stated in the document:
- Formation of a Vast Array of Compounds: Because carbon can bond with itself and many other elements in so many different configurations, it can form an almost infinite number of distinct compounds. This explains the immense chemical diversity observed in nature and in synthetic materials.
- Backbone of Organic Chemistry: This is perhaps the most profound implication. Organic chemistry is the study of carbon-containing compounds. Carbon's ability to form stable, complex molecular frameworks, especially long chains and rings, makes it the literal "backbone" or scaffolding upon which all organic molecules are built. This includes all the molecules essential for life (proteins, carbohydrates, lipids, nucleic acids), as well as countless synthetic materials like plastics and pharmaceuticals.
4. Carbon Allotropes
Finally, the document introduces the concept of allotropes.
- Definition of Allotrope: An allotrope refers to different structural forms in which an element can exist. These forms have distinct physical and chemical properties, even though they are composed of the same type of atom. The differences arise from how the atoms are bonded together or arranged in space.
- Notable Carbon Allotropes:
- Graphite: Characterized by its soft, layered structure, making it useful in pencils and as a lubricant.
- Diamond: Known for its extreme hardness and brilliant luster, a result of its rigid, tetrahedral crystal lattice.
- Amorphous Carbon: A less structured form, often found as soot, charcoal, or carbon black, lacking the ordered crystalline structure of diamond or graphite.
These allotropes beautifully demonstrate how a single element, carbon, can manifest in radically different forms with varied applications, solely due to the arrangement and bonding of its atoms.
For a guided walk-through of the core topics, see the In-depth Study Path.
See also: Timeline of Discoveries, Real-World Applications, Key Terms & Concepts
Home > Glossary of Key Terms
Glossary of Key Terms
Carbon
A fundamental element in the universe, denoted by atomic number six, a non-metal belonging to group 14 of the periodic table, unique for its ability to form stable covalent bonds, and the backbone of organic chemistry.
Fundamental element
A basic and essential element, a description applied to carbon due to its significance.
Atomic number six
The specific number that denotes the element carbon.
Non-metal
A classification for carbon, indicating its type of element.
Group 14
The specific column on the periodic table to which carbon belongs.
Periodic table
The system where elements are organized, showing carbon's position in Group 14.
Covalent bonds
Stable bonds that carbon has a unique ability to form with other elements, including itself.
Compounds
A vast array of substances that carbon forms due to its unique bonding property.
Organic chemistry
The field of chemistry for which carbon is considered the 'backbone'.
Backbone of organic chemistry
Carbon's role as the foundational element that enables the formation of a vast array of compounds.
Allotropes
Different forms in which carbon exists, such as graphite, diamond, and amorphous carbon.
Graphite
One of the notable allotropes of carbon.
Diamond
One of the notable allotropes of carbon.
Amorphous carbon
One of the notable allotropes of carbon.
See also: Summary & Key Points
Home > Timeline of Discoveries
Timeline of Discoveries
1772: Identification of Carbon as an element and Diamond as an allotrope of Carbon
1779: Identification of Graphite as an allotrope of Carbon
- By: Carl Wilhelm Scheele
- Source
1806: Coining of the term "Organic Chemistry"
- By: Jöns Jacob Berzelius
- Source
1828: Synthesis of Urea, marking the beginning of modern organic chemistry
1869: Development of the Periodic Table of Elements
1913: Discovery of the concept of Atomic Number
1916: Development of the Covalent Bond theory
See also: Detailed Study Guide
Home > Real-World Applications
Real-World Applications
(Content for this section was not provided in the source notes.)
See also: Detailed Study Guide
Home > Key Terms & Concepts
Key Terms & Concepts
- Carbon
- Characteristics
- Fundamental element
- Atomic number six
- Non-metal
- Group 14 of the periodic table
- Unique Bonding Property
- Ability to form stable covalent bonds
- With other elements
- With itself
- Consequences of bonding
- Forms a vast array of compounds
- Backbone of organic chemistry
- Allotropes
- Graphite
- Diamond
- Amorphous carbon
See also: Detailed Study Guide
Home > In-depth Study Path
In-depth Study Path
This is a guided path through the core concepts of the document. Start with the first topic and follow the links at the bottom of each page to proceed.
See also: Detailed Study Guide
Home > In-depth Study Path > Carbon: A Fundamental Element's Basic Identity
Carbon: A Fundamental Element's Basic Identity
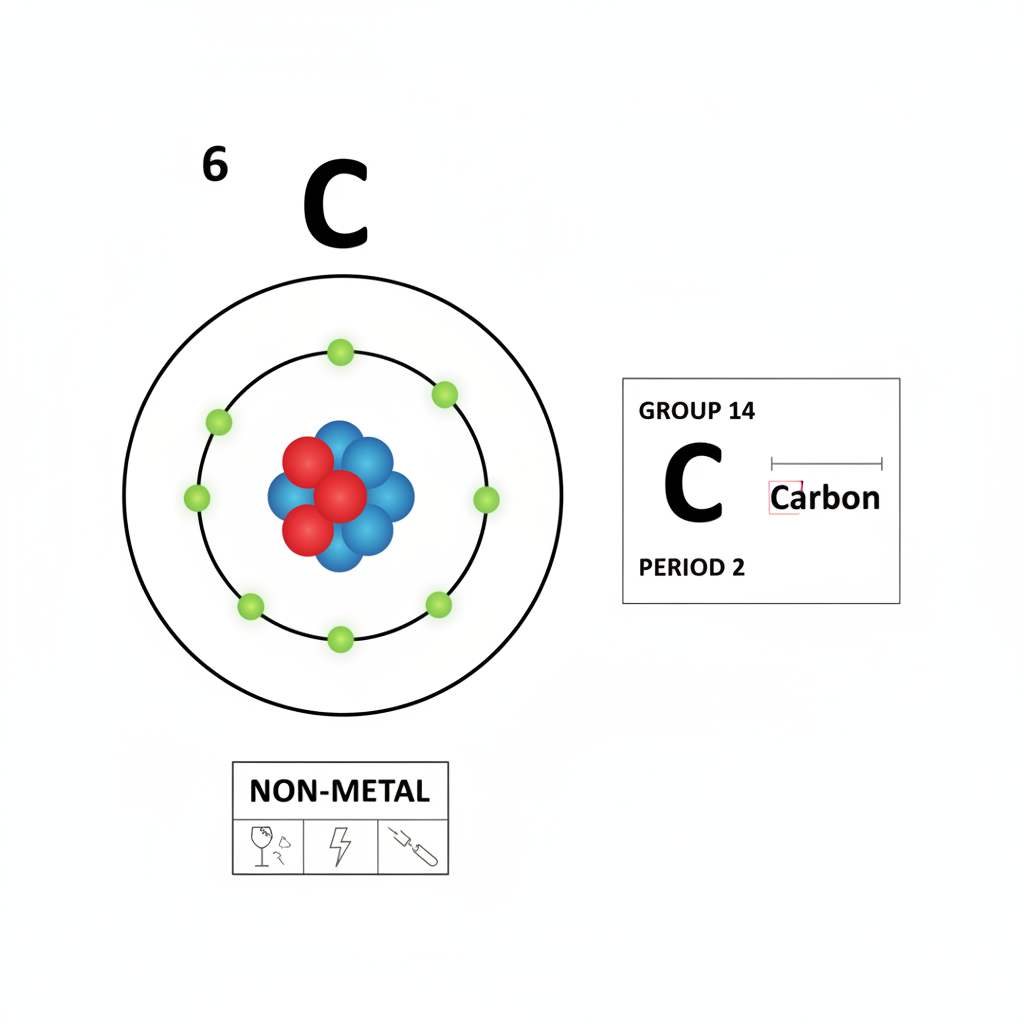
Carbon is introduced as a fundamental element in the universe, highlighting its essential nature. It is identified by its atomic number six, meaning every carbon atom contains six protons. This element is classified as a non-metal, indicating certain characteristic physical and chemical properties distinct from metals. Furthermore, carbon's position in group 14 of the periodic table is specified, providing crucial information about its valence electron configuration and its typical bonding behavior, which usually involves four valence electrons.
➡️ Next: Carbon's Unique Covalent Bonds and Catenation
Home > In-depth Study Path > Carbon's Unique Covalent Bonds and Catenation
Carbon's Unique Covalent Bonds and Catenation
A defining characteristic of carbon is its unique ability to form stable covalent bonds. Covalent bonds involve the sharing of electron pairs between atoms, leading to strong molecular structures. What makes carbon particularly special is its capacity to form these stable bonds not only with other elements (like hydrogen, oxygen, nitrogen) but also extensively with itself. This self-bonding ability, known as catenation, allows carbon atoms to link together in long chains, branched structures, and rings, forming the basis for complex molecules.
⬅️ Previous: Carbon: A Fundamental Element's Basic Identity | ➡️ Next: From Bonding to Diversity: Carbon as the Core of Organic Chemistry
Home > In-depth Study Path > From Bonding to Diversity: Carbon as the Core of Organic Chemistry
From Bonding to Diversity: Carbon as the Core of Organic Chemistry

The extraordinary bonding properties of carbon have profound implications for the diversity of chemical compounds. Its ability to form stable covalent bonds with itself and a wide array of other elements means that carbon can construct an immense and vast array of compounds. This unparalleled versatility is why carbon is recognized as the backbone of organic chemistry. Organic chemistry, the study of carbon-containing compounds, owes its very existence to carbon's capacity to form the intricate and diverse molecular frameworks essential for life and countless synthetic materials.
⬅️ Previous: Carbon's Unique Covalent Bonds and Catenation | ➡️ Next: Carbon's Diverse Manifestations: Allotropes
Home > In-depth Study Path > Carbon's Diverse Manifestations: Allotropes
Carbon's Diverse Manifestations: Allotropes

Beyond its atomic and bonding characteristics, carbon also demonstrates the phenomenon of allotropy. Allotropes are different structural forms of the same element, possessing distinct physical and chemical properties due to variations in how their atoms are arranged and bonded. The document specifically mentions several notable allotropes of carbon: graphite, known for its soft, layered structure; diamond, renowned for its extreme hardness and crystalline lattice; and amorphous carbon, which refers to less structured forms like soot or charcoal.
⬅️ Previous: From Bonding to Diversity: Carbon as the Core of Organic Chemistry
Home > Multiple Choice Quiz
Multiple Choice Quiz
1. What is the atomic number of carbon?
- [ ] Four
- [ ] Six
- [ ] Twelve
- [ ] Eighteen
View Answer & Explanation
Correct Answer: Six
Explanation: The document states that carbon is 'denoted by the atomic number six'.
2. Carbon belongs to which group of the periodic table?
- [ ] Group 1
- [ ] Group 2
- [ ] Group 14
- [ ] Group 18
View Answer & Explanation
Correct Answer: Group 14
Explanation: The document specifies that carbon 'belongs to group 14 of the periodic table'.
3. What type of bonds is carbon uniquely able to form with other elements and itself?
- [ ] Ionic bonds
- [ ] Metallic bonds
- [ ] Hydrogen bonds
- [ ] Covalent bonds
View Answer & Explanation
Correct Answer: Covalent bonds
Explanation: The text states carbon is 'unique for its ability to form stable covalent bonds with other elements, including itself'.
4. Why is carbon considered the 'backbone of organic chemistry'?
- [ ] Because it is the hardest element known.
- [ ] Because it forms a vast array of compounds due to its unique bonding properties.
- [ ] Because it is the most abundant element in the universe.
- [ ] Because it exclusively forms bonds with metallic elements.
View Answer & Explanation
Correct Answer: Because it forms a vast array of compounds due to its unique bonding properties.
Explanation: The document explains that carbon's unique bonding property 'allows carbon to form a vast array of compounds, making it the backbone of organic chemistry'.
5. Which of the following is NOT listed as a notable allotrope of carbon in the document?
- [ ] Graphite
- [ ] Diamond
- [ ] Fullerene
- [ ] Amorphous carbon
View Answer & Explanation
Correct Answer: Fullerene
Explanation: The document lists 'graphite, diamond, and amorphous carbon' as notable allotropes. Fullerene is not mentioned.
Interactive Quiz
Test your knowledge! Select the best answer for each question and submit to see your score.




