As simple as possible
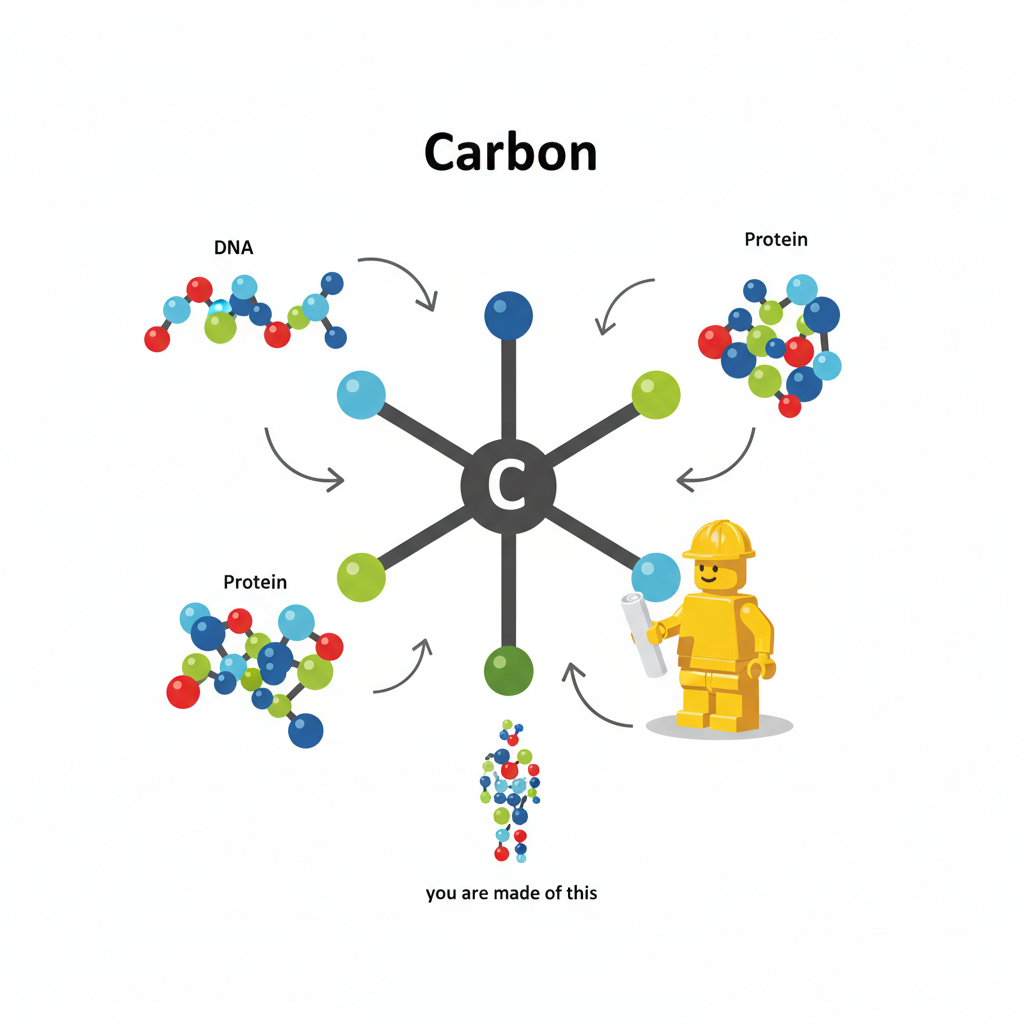
Imagine carbon as the ultimate master builder, like the chief architect or lead LEGO builder for everything that's alive – including you!
Why Carbon is So Important:
Your body, and all living things, are made of huge, complicated molecules (like proteins for your muscles, or DNA for your genetic code). Carbon is the tiny, foundational piece that all these big, essential molecules are built around. It's like the special, most important building block that everything else connects to.
How Carbon Connects:
Every atom wants to be "happy" and stable. Carbon has 4 "sticky spots" (called electrons) in its outer layer, but it wants 8 to be truly happy. So, it always tries to make 4 strong connections (called "bonds") with other atoms by sharing its sticky spots. Think of carbon as a friendly octopus with four arms, always wanting to hold hands with four other atoms to feel complete. This ability to make four strong connections means carbon can create an incredible variety of shapes and structures.
What are Hydrocarbons?
These are the simplest types of carbon-based molecules. They're just carbon atoms holding hands with hydrogen atoms. Methane, the gas you might use for heating or cooking, is a very simple hydrocarbon.
Energy in Hydrocarbons:
The connections (bonds) between the carbon and hydrogen atoms in hydrocarbons are like little energy storage units. When these bonds are broken (like when you burn natural gas), that stored energy is released, usually as heat. So, hydrocarbons are like tiny fuel tanks!
Different Shapes Make Different Things:
Carbon atoms can connect in many ways, like a chain of people holding hands.
- Single Connections: If two carbon atoms connect with just one bond, it's like two people holding one hand each – they can twist and spin around that connection. This makes parts of the molecule flexible.
- Double Connections: If they connect with two bonds, it's like two people holding both hands – they're locked in a flat, stiff position and can't twist.
- Triple Connections: If they connect with three bonds, they're super stiff and straight, like a rigid pole.
Why does shape matter? Because in your body, every molecule has a job, and it needs a very specific 3D shape to do that job, just like a specific key fits only one lock.
Carbon Can Also Make Rings:
Besides chains, carbon atoms can also connect to form closed circles, like tiny rings. Some rings are simple (just single connections), while others have special alternating single and double connections, making them extra stable. These rings are also very important building blocks for many things in your body, like some hormones.