For a Year 10 student: Carbon
Okay, imagine carbon as the ultimate LEGO brick for building everything in living things!
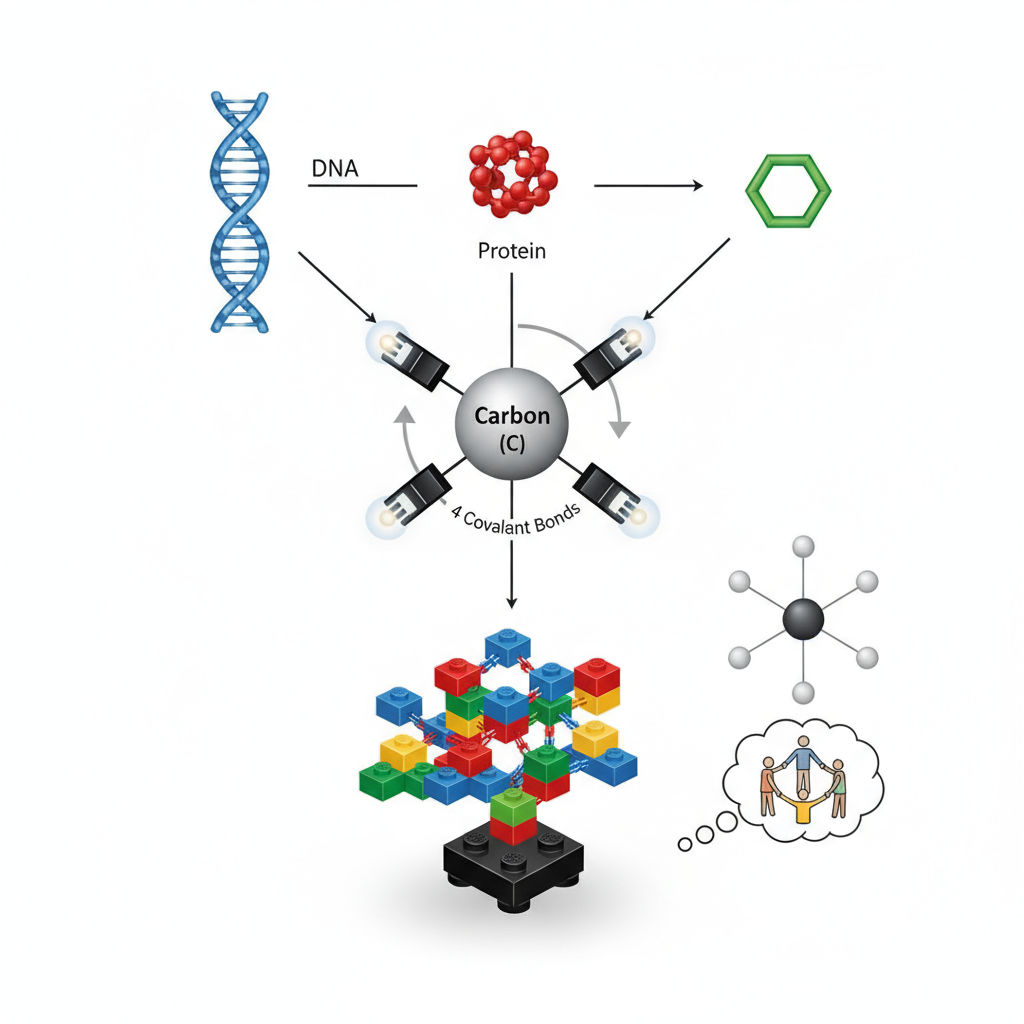
Your body, plants, and all living things are built from huge, complex molecules like proteins (which build muscles) and DNA (which carries your genetic code). Carbon is the central, foundational piece for all these big molecules. Think of it as the main type of LEGO brick you use to build any complicated model.
Why Carbon is so Special
Your body, plants, and all living things are built from huge, complex molecules like proteins (which build muscles) and DNA (which carries your genetic code). Carbon is the central, foundational piece for all these big molecules. Think of it as the main type of LEGO brick you use to build any complicated model.
How Carbon Bonds
Every atom wants to be "happy" and stable by having a full outer shell of electrons. Carbon has 4 electrons in its outer shell but wants 8. So, it's always looking to make 4 strong connections (called covalent bonds) with other atoms by sharing electrons. Imagine carbon as a person with four hands, always wanting to hold hands with four other people to feel complete. This ability to make four strong bonds means carbon can link up with many other atoms in many different ways, making it incredibly versatile.
What are Hydrocarbons?
These are the simplest types of organic molecules, made up only of carbon and hydrogen atoms. Methane, the natural gas you might use for cooking, is a simple hydrocarbon – just one carbon holding hands with four hydrogens.
Energy in Hydrocarbons
The connections (bonds) between atoms in hydrocarbons store a lot of energy. When you break these bonds (like when you burn natural gas), that stored energy is released, usually as heat. So, hydrocarbons are like little energy packs!
Different Shapes and Structures
Carbon can form long chains, like a string of LEGO bricks. These chains can be straight or have branches sticking off them. The way carbons connect also matters:
- Single Bonds: If carbons connect with just one bond, they're like two LEGO bricks connected by a single stud – they can twist and rotate around that connection, making the molecule flexible.
- Double Bonds: If carbons connect with two bonds, they're like two LEGO plates connected side-by-side – they're locked into a flat, rigid shape and can't twist.
- Triple Bonds: With three bonds, they're super rigid and form a perfectly straight line.
This shape is really important! Just like a specific key needs a specific shape to fit into a lock, biological molecules need the right 3D shape to do their job correctly in your body.
Carbon can also form rings, connecting its ends to make circles. Some rings are simple, with only single bonds (aliphatic), while others are special flat rings with alternating single and double bonds (aromatic, like benzene). These unique ring structures are found in important molecules like some hormones.
Test Your Knowledge
See what you've learned about carbon. Choose the best answer for each question.
