Carbon's Essential Role and Bonding Principles
Carbon is the elemental backbone of all organic molecules and macromolecules (proteins, nucleic acids, carbohydrates, lipids) vital for life. Its unique atomic structure makes it ideal for this role:
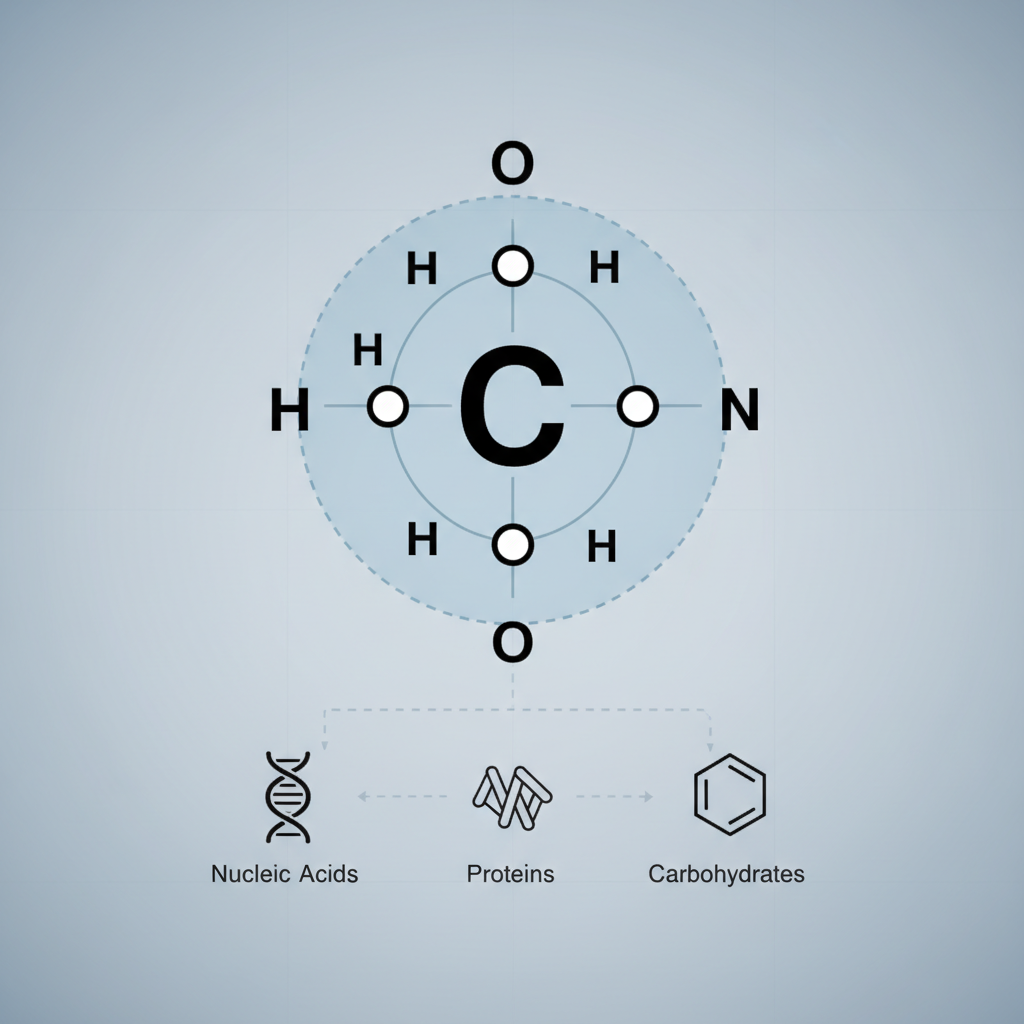
- Electron Configuration: With an atomic number of 6, carbon has 2 electrons in its inner shell and 4 valence electrons in its outermost shell.
- Octet Rule: To achieve a stable electron configuration (like noble gases), carbon seeks to complete its outer shell with 8 electrons. It does this by forming four covalent bonds, sharing electrons with other atoms. This tetravalency is the basis of its versatility.
- Bonding Strength and Flexibility: Carbon forms strong, stable covalent bonds not only with hydrogen, oxygen, nitrogen, and phosphorus but also extensively with other carbon atoms. This allows for the construction of incredibly diverse and complex molecular structures.
- Geometry (sp3 Hybridization): When carbon forms four single bonds, like in methane (CH4), these bonds are oriented as far apart as possible due to electron repulsion, resulting in a tetrahedral geometry with bond angles of 109.5°. This three-dimensional arrangement is crucial for molecular shape.
Functional Groups (Briefly Mentioned)
Although not detailed in the document, functional groups are specific arrangements of atoms (containing oxygen, nitrogen, sulfur, phosphorus, etc.) attached to the carbon backbone. They are critical because they confer specific chemical properties and reactivity to the overall molecule, thus determining its function in biological systems.
Hydrocarbons: Structure, Energy, and Conformation
Hydrocarbons are the simplest organic molecules, composed exclusively of carbon and hydrogen atoms.
- Energy Storage: The numerous C-C and C-H covalent bonds in hydrocarbons store significant amounts of potential energy. This energy is released upon oxidation (combustion), making hydrocarbons excellent fuels (e.g., methane) and their derivatives primary energy sources in living organisms (e.g., fatty acids).
- Hydrocarbon Chains: Carbon atoms can link together to form chains that can be:
- Linear or Branched: Affecting packing and intermolecular forces.
- Single Bonds (Alkanes, e.g., ethane): C-C single bonds allow free rotation around the bond axis. Each carbon is sp3 hybridized, maintaining tetrahedral geometry locally. This rotational freedom leads to a variety of possible conformations for a given molecule.
- Double Bonds (Alkenes, e.g., ethene): C=C double bonds consist of one sigma (σ) and one pi (π) bond. The pi bond restricts rotation, locking the atoms involved into a planar geometry (sp2 hybridization) around the double bond. This fixed arrangement can lead to geometric (cis-trans) isomerism.
- Triple Bonds (Alkynes, e.g., ethyne): C≡C triple bonds consist of one sigma and two pi bonds, enforcing a linear geometry (sp hybridization) for the atoms directly bonded to the triple-bonded carbons.
- Conformation and Function: The specific three-dimensional shape (conformation) dictated by the types of bonds and the arrangement of atoms is critical for biological activity. For example, an enzyme's active site has a specific shape to bind its substrate, and a receptor's binding site interacts only with molecules of a complementary shape.
Hydrocarbon Rings: Aliphatic and Aromatic
Carbon atoms can also form stable ring structures:
- Aliphatic Hydrocarbons: These include linear chains and rings that contain only single bonds between carbon atoms within the ring (e.g., cyclopentane, cyclohexane). These rings can adopt various puckered conformations (e.g., chair and boat forms for cyclohexane) to relieve ring strain, though the document doesn't detail this.
- Aromatic Hydrocarbons: These are characterized by a closed ring of carbon atoms with alternating single and double bonds, forming a conjugated system where pi electrons are delocalized over the entire ring (e.g., benzene). This delocalization confers significant stability (resonance stabilization) to the aromatic system. Aromatic rings are crucial components of many biological molecules, including certain amino acids, steroids (like cholesterol, estrogen, testosterone), and DNA bases. It's also noted that some aromatic compounds, like benzene, are carcinogens.
Test Your Knowledge
Answer the following questions to check your understanding of the material.